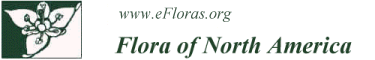Morphology of Mosses (Phylum Bryophyta)
Barbara J. Crandall-Stotler
Sharon E. Bartholomew-Began
With over 12,000 species recognized worldwide (M. R. Crosby et al. 1999), the Bryophyta, or mosses, are the most speciose of the three phyla of bryophytes. The other two phyla are Marchantiophyta or liverworts and Anthocerotophyta or hornworts. The term “bryophytes” is a general, inclusive term for these three groups though they are only superficially related. Mosses are widely distributed from pole to pole and occupy a broad range of habitats. Like liverworts and hornworts, mosses possess a gametophyte-dominated life cycle; i.e., the persistent photosynthetic phase of the life cycle is the haploid, gametophyte generation. Sporophytes are matrotrophic, permanently attached to and at least partially dependent on the female gametophyte for nutrition, and are unbranched, determinate in growth, and monosporangiate. The gametophytes of mosses are small, usually perennial plants, comprising branched or unbranched shoot systems bearing spirally arranged leaves. They rarely are found in nature as single isolated individuals, but instead occur in populations or colonies in characteristic growth forms, such as mats, cushions, turfs, or wefts. While uniform in general life-history features, mosses show extensive morphological and anatomical diversification in both gametophyte and sporophyte organization.
Currently, mosses are classified into five morphologically distinct superclasses (B. Goffinet and W. R. Buck 2004) that are circumscribed as follows: Superclass I, comprising only Takakia; Superclass II, for Sphagnum and Ambuchanania Seppelt & H. A. Crum; Superclass III, with Andreaea and Acroschisma Lindley; Superclass IV, comprising only Andreaeobryum; and Superclass V, all the peristomate mosses, comprising most of the diversity of mosses. Although molecular data have been undeniably useful in identifying the phylogenetic relationships among moss lineages, morphological characters continue to provide definition of systematic groupings (D. H. Vitt et al. 1998) and are diagnostic for species identification. This chapter is not intended to be an exhaustive treatise on the complexities of moss morphology, but is aimed at providing the background necessary to use the keys and diagnostic descriptions of this flora.
Gametophyte Characters
Spore Germination and Protonemata
A moss begins its life cycle when haploid spores are released from a sporophyte capsule and begin to germinate. In the majority of mosses, germination is exosporic, i.e., the spore wall is ruptured by the expanding spore protoplast after its release from the capsule and prior to any cell division. However, in some mosses, e.g., Andreaea, Drummondia, and Leucodon, germination is precocious and endosporic, meaning that cell divisions occur prior to spore release and spore wall rupture, respectively. Such taxa are described as having multicellular spores. Although there are variations in patterns of germination (see K. Nehira 1983), the fundamental product in most mosses is a highly branched filamentous, uniseriate protonema. Cell specialization occurs within the protonema to form two types of filaments, a horizontal system of reddish brown, anchoring filaments, called the caulonema, and upright, green filaments, the chloronema. Each protonema can spread over several centimeters, forming a fuzzy green film over its substrate. Fragments of chloronema, cut out by the formation of specialized abscission cells (tmema), may further disperse the protonema. Usually this protonemal stage is short-lived, but in a few taxa, e.g., Buxbaumia, it persists as the vegetative phase of the plant.
As the protonema grows, target cells usually on the caulonema generate bud initials that will ultimately divide by sequential oblique divisions to form bud apical cells. This initiates the growth of the leafy gametophore or shoot stage of the moss. Usually, numerous shoots develop from each protonema so that, in fact, a single mat or cushion of shoots can be the product of a single spore.
Shoot Morphology and Habit
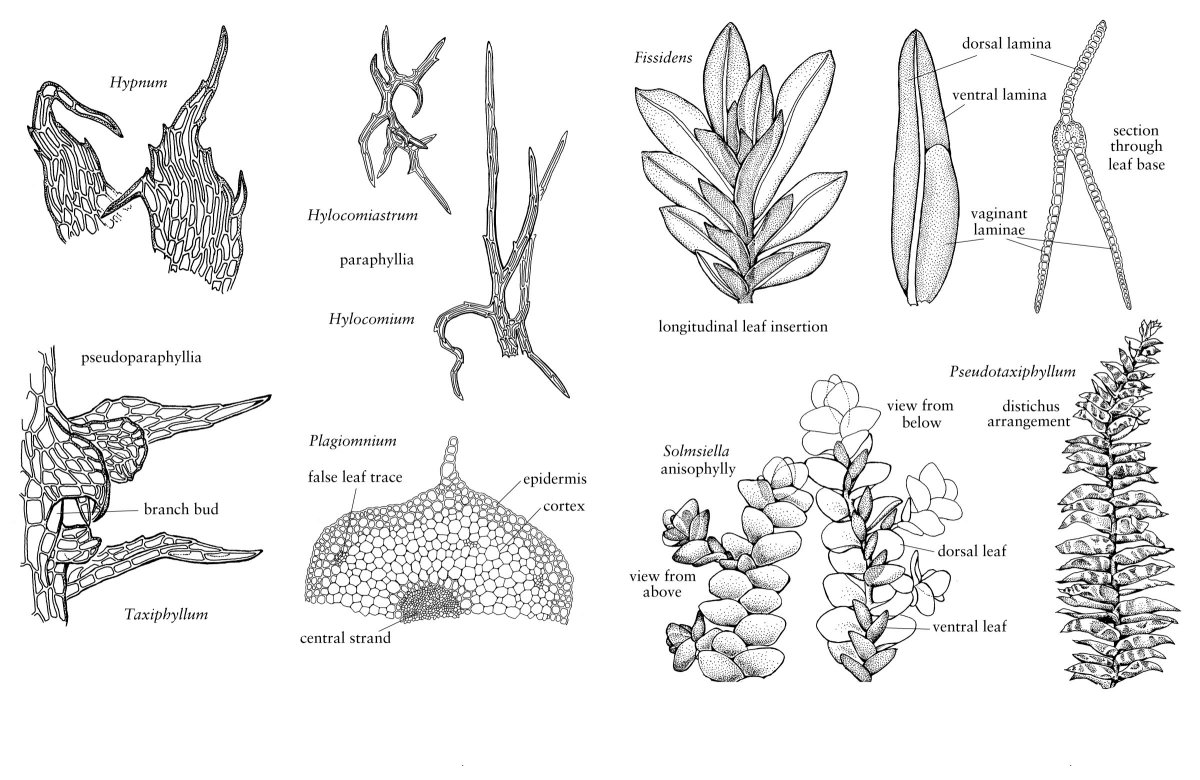
The leafy shoot continues to grow from mitotic activity of its obovoidal to fusiform apical cell and surrounding meristem. Divisions occurring in the apical cell form spirally arranged derivatives, each of which will give rise to a single leaf and a portion of the stem. The angle of divergence between successive derivatives is responsible for the spatial arrangement of the leaves or phyllotaxy of the shoot. In a few mosses, mature leaves are clearly ranked; e.g., the leaves of Fissidens and Bryoxiphium are in two rows, a 1/2 phyllotaxy, and Fontinalis and Tetraphis have leaves aligned in three rows, a 1/3 phyllotaxy. In most mosses, however, the leaves are spirally distributed, with 2/5 and 3/8 phyllotaxies being most common (W. Frey 1971; B. Crandall-Stotler 1984). Branches, which develop from superficial “bud” initials sequestered in the leaf axils, mimic the development of the protonemal buds (J. Berthier 1972) and are generally heteroblastic, meaning that leaves at the base of a branch display juvenile morphology as compared to the later-formed leaves. Especially in pleurocarpic mosses, chloronema- and caulonema-like filaments, termed pseudoparaphyllia and macronemata, respectively, are found at the branch base, further suggesting its budlike attributes (Berthier).
The peristomate or true mosses (Superclass V) have traditionally been divided into two broad morphological groups, namely, acrocarps and pleurocarps, based on the position of the perichaetia and subsequent sporophytes (Fig. 1). Acrocarps are characterized by erect or ascending shoot systems that are either unbranched or only sparingly branched. Branching is typically sympodial with the branches morphologically comparable to the determinant main shoot from which they arise. Perichaetia are differentiated at the tip of the main or primary shoot and terminate its growth, so further plant growth occurs only if a branch is produced below the perichaetium; such branches are called subfloral innovations. Pleurocarps are generally characterized by creeping shoot systems, with extensive lateral branching. In such systems, the indeterminant main stem may be morphologically distinct from the secondary and tertiary level branches that arise from it (C. La Farge 1996). Perichaetia in pleurocarps are produced at the tips of very short, basally swollen lateral branches that are morphologically distinct from the vegetative branches. Because of the extremely reduced size of the perichaetial branches, the sporophytes appear to arise from scattered positions all along the primary stem. Cladocarpic mosses produce perichaetia at the tips of unspecialized lateral branches that display the same heteroblastic leaf series as the vegetative branches. Such branches are themselves capable of branching, and these mosses are neither acrocarpic nor pleurocarpic (La Farge). Although acrocarps, pleurocarps, and cladocarps tend to have different branching architectures, it is the morphology of the perichaetium-bearing module that defines the groups, not branching habit (La Farge).
Pleurocarps form a natural, monophyletic lineage of true mosses (B. Goffinet and W. R. Buck 2004), but cladocarpy has evolved in several different lineages. Acrocarpy, which appears to be the plesiomorphic condition, also characterizes the Takakiopsida, Andreaeopsida, and Andreaeobryopsida. The main stems of Sphagnum (Superclass II) display a furcate or dichotomous branch architecture (H. A. Crum 1984). Along the main stems, fascicles of branches are produced in every fourth leaf (H. Leitgeb 1869), with three or more branches per fascicle. At least two branches in each fascicle hang downwards and are appressed to the stem, while one to three are divergent. Despite their distinctive fascicled arrangements, all branch development in Sphagnum is like that of other mosses, with each branch arising from a single axillary bud initial (Leitgeb). At the apex of the main shoot, the abundant developing fascicles are tightly clustered into a dense tuft called the capitulum. Archegonia terminate special, short branches in the capitulum.
Rhizoids
Except for Takakia and Sphagnum, mosses are anchored to their substrates by filamentous, often branched, reddish brown rhizoids. As in caulonemata, the rhizoids are multicellular with oblique cross walls; their walls are smooth or roughened with papillae. Rhizoids can be restricted to the base of the stem, especially in acrocarps, or arise all along the stem where it is in contact with the substrate. In some taxa, e.g., Tomentypnum, densely packed rhizoids form a feltlike tomentum on the stem. Most rhizoids are slender and only sparingly branched (micronematal type) but others are larger in diameter and extensively branched (macronematal type). The former arise from any of the epidermal cells of the stem, but the latter type is associated only with branch primordia as discussed earlier. Rhizoids are not major sites of water and nutrient uptake, but can enhance capillary movement of water along the outer surface of the stem (M. C. F. Proctor 1984). They function primarily as anchoring structures and in some taxa, e.g., Leptobryum and Pyramidula, form asexual propagules that aid in the spread of the colony over unoccupied substrate.
Stem Anatomy
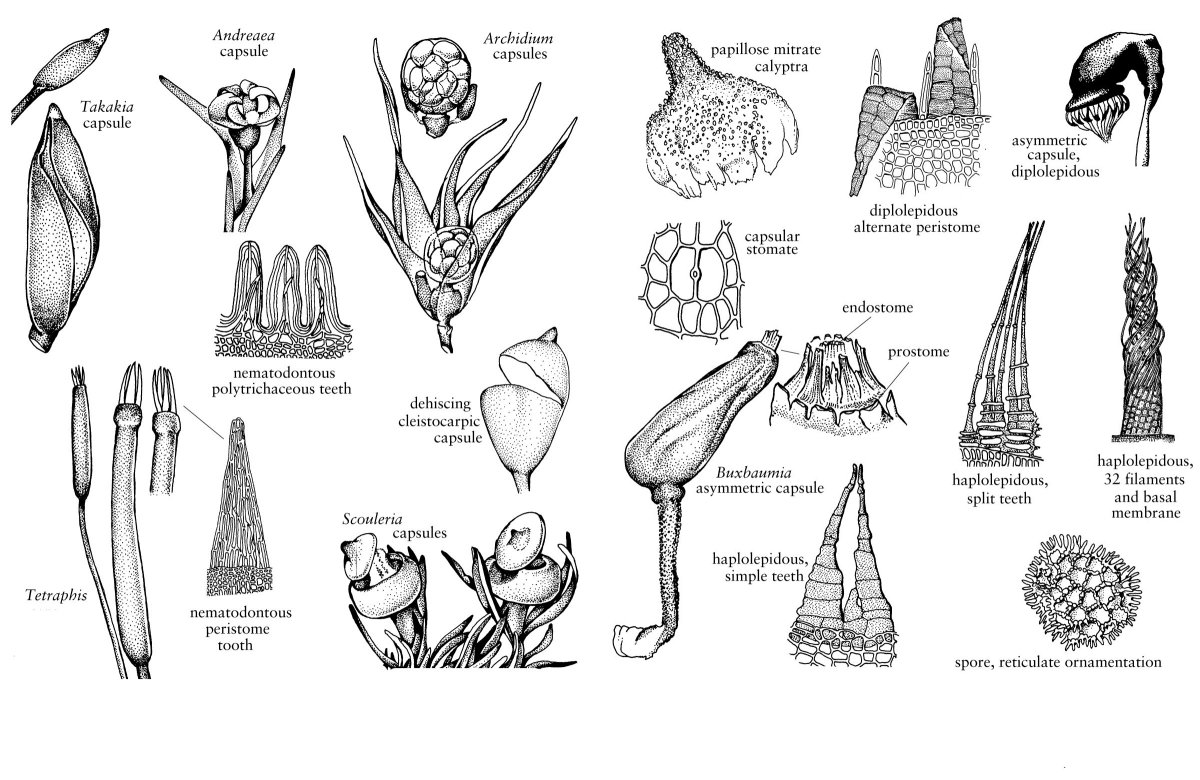
In many mosses, the stem is anatomically complex, consisting of a differentiated epidermal layer, a cortex, and a central strand of thin-walled, hydrolyzed water-conducting cells, called hydroids (Fig. 2). The epidermal cells are typically elongate in surface view and have thick, pigmented walls and small lumina. Various types of waxy deposits, analogous to cuticle or epicuticular waxes, may cover the surface of the epidermal cell wall, in both stems and leaves, especially in endohydric mosses (M. C. F. Proctor 1979). When these waxes are abundantly produced, the plants appear glaucous or iridescent to the naked eye. Although a thick-walled epidermis is common, in a variety of taxa, e.g., elements of the Pottiaceae as well as taxa of very wet habitats like Hygrohypnum, epidermal cells are thin-walled and swollen, forming a unistratose hyalodermis (Fig. 1). The cortex is generally divided into two zones. The outer zone, or sclerodermis, just to the inside of the epidermis, is comprised of stereids. These are elongated, thick-walled cells, generally with living protoplasts, that function in mechanical support, analogous to collenchyma in vascular plants. The inner zone of the cortex, or central cylinder, contains photosynthetic parenchyma cells that become starch-filled and hyaline toward the stem interior. Some of the cells of the inner cortex are conducting parenchyma that are responsible for the transport of assimilates. The central strand, absent in some taxa, is always a solid core of nonlignified, water-conducting hydroids. Hydroid structure varies among taxa (C. Hébant 1977), from very elongate cells with thickened lateral walls and partially hydrolyzed end walls in the Polytrichopsida to the moderately elongate, small diameter, thin-walled hydroids of most taxa. In addition, the Polytrichopsida have a central strand of hydroids, or hydrome, surrounded by a dissected or medullated ring of highly differentiated, phloemlike sieve cells, called leptoids. These more specialized cells function in the conduction of photoassimilates in concert with the elements of conducting parenchyma; they are of widespread occurrence in immature sporophyte setae, but in the gametophyte generation are restricted to the Polytrichopsida.
|
|
FIGURE 2. Stem anatomy, paraphyllia, pseudoparaphyllia and variations in leaf morphology and arrangement. |
In some taxa, groups of hydroids extend from the leaf costa into the parenchymatous zone of the cortex (Fig. 2). These small clusters of hydroids may remain only in the cortex as false leaf traces, e.g., Plagiomnium, or may extend through the cortex to connect with the central strand as true leaf traces, as occurs in the Polytrichopsida. There is substantial variation in stem organization that is systematically useful, such as number of cell layers in the sclerodermis, depth of the photosynthetic zone, and presence or absence of the central strand and/or leaf traces (I. Kawai 1989). Sphagnum possesses a unique stem anatomy in which the epidermis consists of one to five layers of large cells that are dead at maturity; in some species these cells bear fibril thickening bands, and on the divergent branches they may be modified to form retort cells. The living cells to the inside of the epidermis are differentiated into an outer zone of elongate cells with thick, often pigmented walls (the woody cylinder) and an inner core of large, thin-walled cells.
Variation also occurs in the formation of external structures that are associated with stems. The stem surface may be smooth, or occasionally papillate, or especially in pleurocarps, be covered with green, filamentous, or sometimes foliose paraphyllia (Fig. 2). These small outgrowths presumably photosynthesize and probably enhance water conductivity by capillary action along the stem. Pseudoparaphyllia are additional structures found only in pleurocarps. Although structurally similar to paraphyllia, pseudoparaphyllia are formed only at the bases of branches. They likely serve to protect the branch primordia with which they are associated.
Leaves
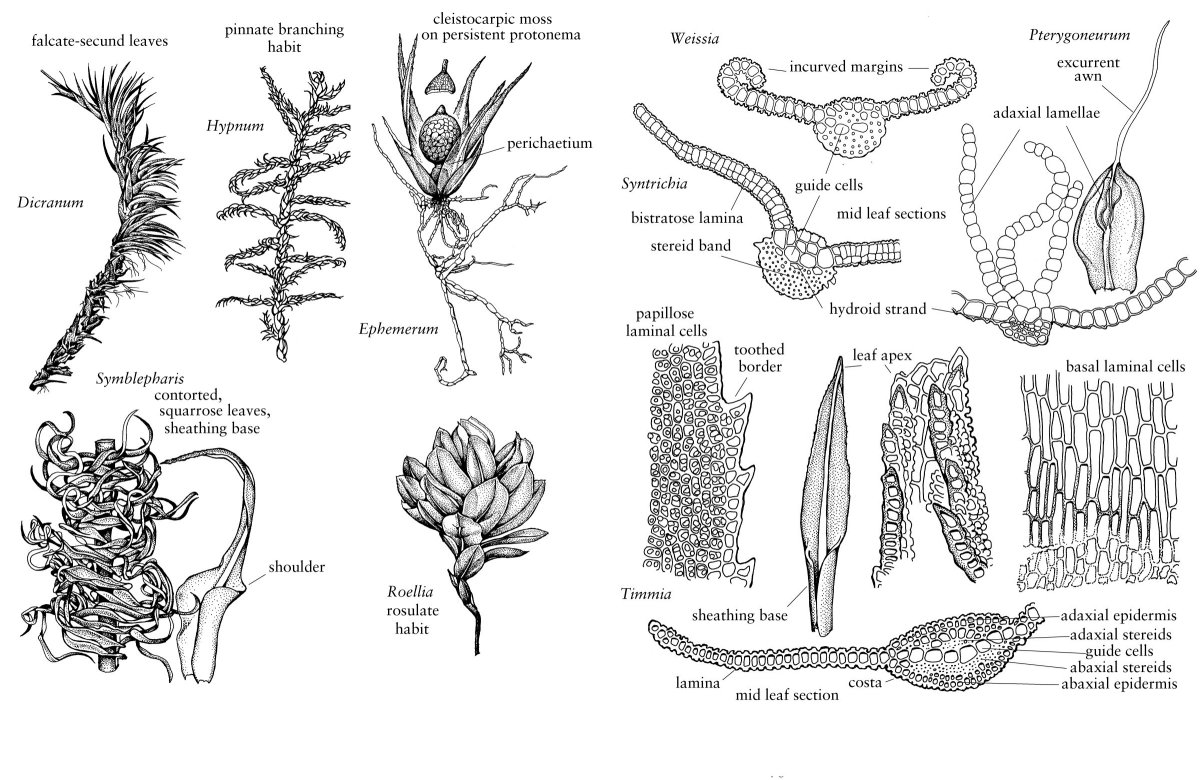
The considerable variation that occurs in the arrangement and structure of moss leaves provides some of the most useful characters for species identification (Figs. 2, 3). Leaves typically arise from all sides of the stem, most commonly exhibiting a spiral phyllotaxy, but distichous and tristichous arrangements can also be found. The mature leaves of a given shoot are usually all similar in size and shape, i.e., isophyllous, but there are taxa that are anisophyllous, with either dorsal or ventral leaves decidedly smaller than the lateral leaves (Fig. 3). Except for a few taxa like Fissidens, leaves are attached to the stem along broad transverse lines. They are generally oriented so their apices diverge radially outward from the stem. In erect mosses, this insertion exposes the adaxial surface of the leaf, that surface directed toward the stem, to light from above and directs the abaxial surface down toward the substrate. In prostrate mosses, in contrast, the adaxial surface is directed toward the substrate, and the abaxial surface is exposed. For this reason, in many keys and descriptions the abaxial surface is defined as the dorsal side of the leaf, and the adaxial surface, as the ventral side (Fig. 1). Sometimes surface features are different on the two sides of the leaf, e.g., one surface may be smooth and the other papillose. In some taxa, all of the apices of the leaves curve to the same side in a secund arrangement (Fig. 3). In others, they may be squarrose, meaning the apices diverge outward, then bend abruptly downward; or julaceous, meaning they are strongly concave on the shoot; or complanate, meaning the adaxial surface of the leaf lies against the stem to give the shoot a flattened appearance.
|
|
FIGURE 3. Variation in leaf morphology, anatomy and habit. |
Moss leaves are undivided and are typically lanceolate to ovate, except for Takakia, which has unique leaves that are two to four times divided into cylindrical lobes. The fundamental moss leaf consists of a unistratose lamina and a multistratose costa or midrib (Fig. 3). However, in a few families, the entire leaf is multistratose, and in many others, leaf margins are multistratose, or otherwise differentiated from the rest of the lamina (Fig. 4). For example, often the margin bears a border of hyaline, elongated cells with thickened walls. Such marginal borders, termed limbidia (singular limbidium), are thought to provide additional support to the lamina. Leaf margins may be plane, recurved, or incurved and are often toothed, with the teeth varying from pointed cellular protuberances to large, several-celled projections.
The areolation or cellular network of the leaf lamina also provides useful characters in moss systematics (Fig. 4). Laminal cells in acrocarps are often short and isodiametric, particularly in the distal portion of the leaf, while the typical pleurocarp bears laminal cells that are long and thin; however, all cell shapes, from linear to oblate, can be found in either group. Usually, cell shapes and sizes are not uniform throughout the leaf. In particular, the cells near the leaf base are comparatively longer and broader than those of the leaf middle, and still smaller cells occur at the leaf apex. The basal cells of the leaf margin, known as alar cells, are also frequently differentiated in size, shape or color from the other cells of the lamina (Fig. 1). Sometimes this zone of differentiated cells extends from the margin all the way to the costa. Hyaline alar cells that are inflated and thin-walled may control leaf orientation in response to hygroscopic conditions, causing the leaf to collapse downward during dry conditions. Laminal cells are smooth or variously ornamented, commonly bearing a variety of wall projections called papillae (Fig. 3). Papillae are heterogeneous in form, size, and distribution. They may be solid or hollow, simple or branched, single or multiple, on both exposed cell surfaces or only on the abaxial surface, over the cell lumen or just at the distal (occasionally proximal or both) end of the cell (prorose or prorulose). Various types of epicuticular wax deposits may add to this ornamentation (M. C. F. Proctor 1979).
The costa or midrib of the leaf provides yet another suite of taxonomically useful characters (Fig. 3). Leaves of acrocarps usually have a clearly defined, single costa that extends from the base of a leaf to shortly before its apex, or to its apex (percurrent) or even beyond its apex (excurrent). In a few acrocarps and many pleurocarps, the costa is short, generally confined to the leaf base, and double, or absent (Fig. 1). Costa anatomy is variable with several distinctive patterns (I. Kawai 1968). The costa may be homogeneously composed of thick-walled stereids, or have adaxial or abaxial epidermal cells larger than the internal cells. Often, a row of large, thin-walled cells, referred to as guide cells, or deuters, bisect the costa, extending between the two laminar segments; occasionally, there will be a small strand of hydroids between the guide cells and one of the stereid bands, especially in those acrocarpic taxa in which leaf traces occur. As suggested by the abundance of stereids in most costae, one of the primary functions of the costa is to provide support for the unistratose lamina, but it can also function in transport. The guide cells of the costa are conducting elements involved in the symplastic transport of photoassimilates from the leaf lamina into the stem cortex (C. Hébant 1977), and the hydroid strands move water upward from the stem into the distal portions of the leaf.
In the Polytrichopsida, as well as a few other taxa, unistratose, photosynthetic lamellae arise from the adaxial (ventral) surface of the costa. They extend as parallel sheets of cells from just beyond the sheathing leaf base often to the apex of the costa. In some taxa, the lamina is incurved over the lamellae, thereby forming an almost mesophyll-like tissue. Variations in the number and height of lamellae as well as lamellar cell morphologies provide taxonomically informative characters in this group. Leaves of the Fissidentaceae are also distinct from those of other mosses, consisting of two basal, clasping vaginant laminae and two vertically oriented, distal laminae, designated as the ventral (superior) lamina and dorsal (inferior) lamina. The two distal laminae arise from the adaxial (ventral) and abaxial (dorsal) surfaces of the costa, respectively, while the vaginant laminae extend laterally from the costa as in other mosses. Still another modification in leaf organization occurs in Sphagnum and several other taxa, e.g., Leucobryum and Leucoloma, in which the leaf is composed of two types of cells, very small, chlorophyllose cells (chlorocysts) and empty, hyaline cells (hyalocysts in Sphagnum or leucocysts in other taxa). In Leucobryum, the leaf is multistratose, with the chlorocysts embedded beween the two layers of leucocysts. In Sphagnum the chlorocysts form a reticulum within a unistratose leaf; variations in pore distribution in the hyalocysts and arrangement of chlorocysts in relation to hyalocysts provide important taxonomic characters in this genus.
Juvenile moss leaves possess one to several uniseriate hairs in their axils. These are persistent in some acrocarps, but are usually physiologically active and secrete mucilage only near the shoot apex. They often deteriorate as the leaf matures. Variation in the number of hairs per leaf and the number and length of cells per hair, nonetheless, has proven useful in some moss groups (L. Hedenäs 1989).
Asexual Diaspores
It has been estimated that up to 15% of moss taxa produce some type of asexual diaspore (H. A. Crum 2001). These include caducous leaves, stems, and rhizoids, as well as morphologically specialized brood bodies. Fragments of gametophytes, if dispersed to a suitable substrate, produce protonemata from which new shoots will develop. Brood bodies are of two major types, gemmae, which are small, unicellular or more commonly, multicellular structures comprised of undifferentiated cells, and propagula, which are small, easily detached plantlets or buds (Fig. 4). Gemmae can be filiform, discoid, or cylindrical, and often possess pigmented and thickened outer cell walls. They can be produced in special splash cups, as in Tetraphis, or near the tip of elongate shoot apices (e.g., Aulacomnium), in the axils of leaves (e.g., some species of Didymodon), or on the laminar or costal surfaces (e.g., Orthotrichum), as well as on rhizoids and protonemata (e.g., Zygodon). Rhizoidal gemmae that are somewhat buried are commonly referred to as tubers. Propagula, sometimes termed bulbils, may be clustered at the tips of the shoots as in Platygyrium, or occur singly in the axils of leaves as in Leptobryum.
Sexual Reproduction
Gametangia are typically clustered with interspersed, sterile hairs, called paraphyses, at shoot or branch apices. Androecia, or male inflorescences, contain numerous, elongate, ovoid to cylindrical antheridia (over 100 in some taxa) and paraphyses, surrounded by perigonial leaves, and may be either budlike or disciform. Gynoecia, or female inflorescences, contain groups of long-necked, stalked archegonia, paraphyses, and surrounding perichaetial leaves, and are only budlike, never disciform. In rare cases, e.g., Takakia, organized androecia and gynoecia are lacking and the gametangia simply occur in the axils of unmodified leaves. Both the organization and arrangement of androecia and gynoecia are of taxonomic importance.
Mosses are either dioicous, i.e., bearing androecia and gynoecia on separate gametophytes, or monoicous, bearing both sexes on a single gametophyte. There are several different kinds of monoicous arrangements, depending on the relative positions of the antheridia and archegonia. In autoicous arrangements, there are separate androecia and gynoecia on the same plant, often on separate branches (cladautoicous), while in both synoicous and paroicous arrangements antheridia and archegonia occur in a single inflorescence, either intermixed within the same cluster (synoicous), or in separate clusters in different leaf axils (paroicous). In plants described as heteroicous, more than one form of monoicy occurs on the same plant. In a few dioicous mosses, e.g., Dicranum, male plants are highly reduced and actually grow as epiphytes upon the leaves or stems of the much larger female plants. Although physiologically dioicous, the production of dwarf males to some degree mimics the male/female relationship of autoicous taxa, so this is sometimes called a pseudautoicous condition. When male plants arise from the same protonema as female plants, it mimics dioicy, and this feature is termed rhizautoicy.
Numerous biflagellated sperm are produced by mitosis inside the antheridia while a single egg develops in the base of each archegonium. When the sperm mature, the antheridia swell and open at their apices. Drops of rain water or morning dew disperse the sperm to a gynoecium, perhaps aided by microarthropods (N. Cronberg et al. 2006). Slimy mucilage secretions in the archegonial necks help pull the sperm downward to the egg. Fertilization of an egg initiates the diploid sporophyte phase.
Sporophyte Characters
Embryo Development
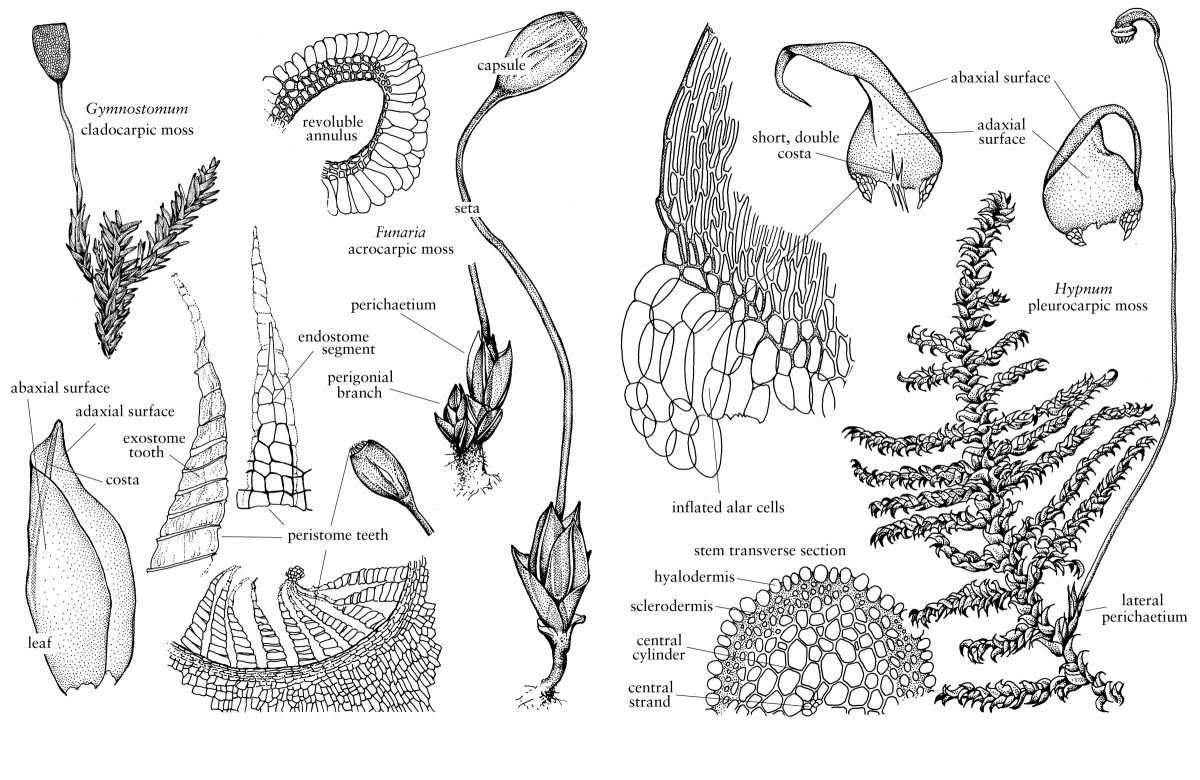
Concomitant with growth of the embryonic sporophyte, cell divisions in the surrounding archegonial center, basal archegonial stalk, and subtending gametophyte shoot produce an enclosing epigonium. Early in development, a nutrient transfer zone, or placenta, is differentiated between the conical foot of the sporophyte and the basal part of the epigonium. Both organic nutrients and water move from the gametophyte into the sporophyte across the placenta. A stemlike seta is differentiated above the foot and embryonic capsule initials are cleaved from an apical cell at the apex of the sporophyte. The establishment of a seta meristem just below the differentiating capsule elongates the seta, tearing the epigonium. The basal part of the epigonium, which still encloses the lower part of the seta and foot, is now referred to as the vaginula, and the upper part that remains over the tip of the sporophyte is the calyptra. In taxa in which the seta is lacking or much reduced, such as Sphagnum and Andreaea, the enlarging capsule ruptures the epigonium. In a few mosses, e.g., Bryobartramia, the epigonium remains intact, never separating into a vaginula and calyptra (H. A. Crum 2001, fig. 25A). Variation in the timing of epigonial tearing can affect calyptra morphology; for example, calyptrae torn away late in development are elongate, as in Encalypta, while those separated early are short and thin, as in Archidium.
The calyptra remains seated over the developing capsule until spore maturation is complete. Calyptrae are of two types based on form. The cucullate type is slit up one side and sits like a hood over the capsule, and the mitrate type is conic and undivided or equally lobed at the base. Variations in calyptra shape, size, areolation, and surface ornamentation provide taxonomically useful characters, as detailed by P. Janzen (1916).
Seta Anatomy
The seta and capsule continue to develop after the emergence of the sporophyte from the epigonium, the seta from a generalized, apical meristem and the capsule by patterned divisions in earlier formed capsule initials. Usually only one sporophyte matures per gynoecium, but in some taxa, e.g., Dicranum, more than one can develop, resulting in polysety, or multiple setae emerging from a single perichaetium.
Anatomically, the seta resembles the stem of the gametophyte in being differentiated into epidermis, cortex of outer stereid and inner parenchymatous zones, and a central conducting strand. There is always a waxy, cuticlelike covering associated with the epidermis. Central strands can be well-developed even in taxa that lack such strands in their gametophytes, and residual leptoids are reported to occur in some taxa, e.g., Funaria, Splachnum, and Meesia, although there is disagreement as to the exact nature of these cells (D. Schulz and C. Wiencke 1976; C. Hébant 1977). Prior to capsule ripening, the seta is typically chlorophyllose, but at maturity it becomes reddish or yellowish brown, due to pigments deposited in the thickened walls of epidermal cells and stereids. It is not uncommon for the interior cells of the seta, including those of the central strand, to deteriorate in late stages of spore maturation, producing a hollow seta (W. Lorch 1931; Hébant). In most mosses the seta surface is smooth, but in some taxa it is roughened with papillae or bristly outgrowths. Often, the epidermal cell rows are helically aligned; this arrangement as well as differential wall thickness on different sides of the seta allows the seta to twist as it dries, and untwist when rehydrated. The direction and pattern of the twist is taxonomically useful in some groups. The extent of seta emergence above the perichaetium varies among taxa. In the Sphagnopsida and Andreaeopsida the seta is absent, and the capsule is instead elevated above the perichaetium by elongation of the gametophytic cells subtending the foot. This extended gametophytic tissue is called a pseudopodium.
Capsule Anatomy
Mosses are monosporangiate, meaning that each sporophyte produces only a single sporangium, or capsule. Very early in development, periclinal divisions in the apically produced capsule initials separate an inner, endothecial zone from an outer ring of amphithecial cells. With the exception of Sphagnum, a columella and spore-producing archesporium are formed from the endothecium, and outer parts of the capsule, including the peristome, develop from patterned divisions of the amphithecium. Details of these later stages in capsule ontogeny have been documented by several workers, including, among others, F. Kienitz-Gerloff (1878), J. C. French and D. J. Paolillo (1975), S. R. Edwards (1984), and A. J. Shaw et al. (1989).
Variations in the morphology of the mature capsule provide a wealth of systematically important characters (Fig. 5). Capsules vary in shape from spheroid to ovoid, obovoid, pyriform, turbinate, ellipsoid, or long-cylindric, and can be either symmetric, e.g., Orthotrichum, or asymmetric, e.g., Funaria. They may be erect, inclined, nodding (cernuous), or even pendent. They are typically terete in transverse section, but can also be 4-angled, e.g., members of the Polytrichopsida, or longitudinally furrowed, with 8 or 16 ribs. The exothecium, or epidermis, of the capsule bears a shiny cuticlelike layer, and is usually smooth-surfaced, although surface ornamentations occur in a few genera. Exothecial characters, such as cell size, shape, and arrangement can be of systematic value. Immature capsules are green, but when mature are various shades of yellow, red, or brown.
Distinctive types of capsule anatomy characterize the five major groups or superclasses of mosses. In the majority of true mosses (Superclass V), capsules consist of three anatomically distinct zones, namely, the basal neck, the median spore-containing urn or theca, and the distal operculum. Capsules with a dehiscent operculum are stegocarpous. Capsules that lack an operculum and dehisce irregularly due to generalized breakdown of the capsule walls are cleistocarpous. In Takakia (Superclass I), Andreaea (Superclass III), and Andreaeobryum (Superclass IV), there is no differentiation of a neck or operculum; their capsules open along incomplete longitudinal sutures, with the capsule staying intact above and below the slits (Fig. 5). There is one spiral suture in Takakia and four vertical sutures in Andreaea and Andreaeobryum. The globose capsule of Sphagnum (Superclass II) possesses a small, lidlike operculum, but lacks a neck; in contrast to true mosses its spore mass, or archesporium, overarches a dome-shaped columella, an arrangement that is also characteristic of Andreaea and Andreaeobryum. In Sphagnum, contraction of the capsule wall releases the operculum, explosively dispersing the spores.
In some mosses, the neck tapers gradually into the seta, but in others, it is markedly differentiated, as a swollen hypophysis (or apophysis), the most notable of which is the highly inflated, parasol-like hypophysis in the Splachnaceae. Stomatal complexes, i.e., stoma and their associated guard cells, are frequent within the exothecium of the neck; these may be superficial or sunken (immersed) in different species of the same genus, e.g., Orthotrichum, and can be either open or closed. The guard cells are usually completely divided, as in vascular plants, but in some taxa, like Funaria, they are only partially divided so that the stoma appears as a slitlike opening in a single cell. In Sphagnum, pairs of guard cells occur scattered throughout the exothecium, but there are no stomata associated with them. Variations in number, form, and distribution of stomata are taxonomically informative (J. A. Paton and J. V. Pearce 1957).
Internally, the central strand of the seta extends up into the neck; this strand is surrounded by a chlorophyllose zone of either somewhat spongy parenchyma or highly differentiated aerenchyma, into which the stoma open. This is a major site of photosynthesis in the developing capsule, providing up to 50% of the photoassimilate necessary for capsule growth (M. C. F. Proctor 1977).
A solid column of parenchymatous cells forms the central columella that extends from the neck through the urn to the operculum of the capsule. The often elongate spore sac encircles the columella. To the outside of the spore sac, the wall of the capsule is differentiated into an inner aerenchyma of air spaces transversed by chlorophyllose filaments, an outer zone of hyaline cells and the thick-walled exothecium. In Polytrichopsida, a filamentous aerenchyma can also be formed between the spore sac and the columella.
The operculum of stegocarpous mosses is attached to the distal end of the urn by an annulus, i.e., a ring of differentiated cells that tear to release the operculum during capsule dehiscence. Annulus anatomy varies among taxa, in both the number of cell layers and the structure of the cells comprising it. In its simplest form, the annulus consists of a ring of small, quadrate cells, often several cells high, that differ in wall thickness from the neighboring exothecial and opercular cells. This type of annulus is either persistent or falls off in fragments when the capsule is moistened due to differential swelling of its cells compared to the neighboring cells. This is the common mode of capsule dehiscence in pleurocarps, but also occurs in other taxa, e.g., Orthotrichum. In many acrocarps, e.g., Funaria and Bryum, the annulus consists of an internal ring of enlarged cells with hydrophilic walls and an outer ring of small cells with thickened, hydrophobic walls (Fig. 1). During dehiscence, the internal cells swell with the uptake of water, but the outer cells remain unchanged. This differential swelling causes the annulus to coil away in a single piece from the overlapping cells of the operculum; this is termed a revoluble annulus. In either case, removal of the operculum, or capsule lid, exposes the mouth of the urn and its spore mass for dispersal.
The Peristome
In the majority of stegocarpous mosses, spore dispersal is mediated by the peristome, a circular system of teeth that is inserted on the mouth of the urn, to the inside of the operculum. The developmental history and architecture of the peristome provide a suite of important systematic characters (Fig. 5). Peristomes are of two fundamentally different types, nematodontous, which are found only in Polytrichopsida and Tetraphidopsida, and arthrodontous. In a nematodontous peristome, the teeth are constructed of bundles of whole, dead cells. Commonly in the Polytrichopsida, 32 or 64 (rarely 16) short lingulate teeth, comprised of up to four layers of vertically elongate, very thick-walled cells, are attached by their inner surface to a membranous expansion of the columella called the epiphragm. The release of the operculum exposes small slits between the teeth through which the spores are slowly released. In the Tetraphidopsida, there are four erect, wedge-shaped peristome teeth, each of which represents a quadrant of the peristomial cell layers.
In contrast to the cellular peristomes of these taxa, arthrodontous peristomes, found in the rest of stegocarpous mosses, consist at maturity only of remnants of paired, periclinal cell walls. As reviewed by several authors (e.g., S. R. Edwards 1984; A. J. Shaw and H. Robinson 1984; W. R. Buck and B. Goffinet 2000), arthrodontous peristomes differentiate from the three innermost layers of the amphithecium formed by fundamental square divisions (K. Goebel 1900–1905, vol. 2) in the apex of the embryonic capsule. Following H. L. Blomquist and L. L. Robertson (1941), these are termed the outer peristomial (OPL), primary peristomial (PPL), and inner peristomial layers (IPL). The number of cells in the peristomial layers in a 1/8 slice of a transverse section is expressed as the peristomial formula (Edwards 1979); thus, a peristomial formula of 4:2:3 describes a capsule with 32 OPL, 16 PPL, and 24 IPL cells. The number and arrangement of cells in the peristomial layers cannot always be determined with certainty in mature capsules, so peristomial formulae are generally not included in taxonomic descriptions.
Arthrodontous peristomes are of two major types, namely, haplolepidous and diplolepidous (Fig. 5). The haplolepidous peristome consists of a single ring of 16 teeth that are formed by cell wall deposition on the paired walls of the PPL and IPL. The peristomial formula is always 0(4):2:3, with a single column of PPL cells forming the outer (dorsal) surface of each tooth, and unequal parts of two IPL cells forming the inner (ventral) surface. Consequently, the outer surface of the tooth, which may be variously ornamented with horizontal striae, trabeculae, or papillae, lacks median or divisural lines (= vertical cell walls). The teeth can be forked at their apices, as in the Dicranaceae, or be fused at the base into an elongate tube, or basal membrane, or be divided into 32 long narrow, filaments, e.g., the Pottiaceae. Development from the OPL is highly reduced or absent, forming at best prostomial bumps at the base of the peristome (S. R. Edwards 1984).
Diplolepidous peristomes have the same number of cells in the OPL and PPL as haplolepidous peristomes, but display substantial variation in the IPL numbers, with peristomial formulae ranging from 4:2:4 to 4:2:14. Two sets of teeth are differentiated, the exostome, or outer peristome, formed by deposition on the paired walls of the OPL and PPL, and the endostome, formed at the PPL–IPL wall junctures. The exostome typically consists of 16 teeth, equal to the number of cells in the PPL, while the outer surface of each tooth bears a divisural line that marks the two columns of cells of the OPL. The teeth may be joined together in pairs, or secondarily divided, and are often highly ornamented, especially on the outer surface (A. J. Shaw 1985). The architecture of the endostome is likewise variable, with different patterns of surface ornamentation on outer and inner surfaces (Shaw and J. R. Rohrer 1984). In a diplolepidous-alternate peristome (D. H. Vitt 1984) of the bryoid or hypnoid type, the endostome comprises a basal, often keeled membrane, topped by 16 broad, perforate segments that alternate with the exostome teeth. One to four uniseriate cilia occur between the segments, opposite the exostome teeth. In some taxa, the endostome segments are highly reduced or absent, and the inner peristome consists only of cilia (Fig. 5). In contrast, in the diplolepidous-opposite peristome of the Funariales, there is no basal membrane, the endostome segments occur opposite the exostome teeth, and there are no cilia (Fig. 1). In some taxa, e.g., Orthotrichum, a short, rudimentary system of processes, called a prostome or preperistome, is formed just to the outside of the outer teeth.
Movements of the exostome teeth of diplolepidous taxa as well as the single ring of teeth of haplolepidous taxa are due to the differential composition of the wall deposits on the outer versus the inner surfaces of the teeth. Specifically, one surface readily absorbs water and elongates, while the other does not. This differential response to water absorption causes the teeth to bend when moistened. In many taxa the teeth close over the mouth of the capsule when moistened, so spores are released only when the air is dry, but in others they bend outward when wet, allowing spore release in moist conditions (D. M. J. Mueller and A. J. Neumann 1988). With drying, the teeth return to their original stance. This process can be repeated several times, resulting in the gradual release of the spores from the capsule.
Arrest of peristome development can result in the loss of segments, cilia, teeth, the entire endostome or exostome, or the whole peristome. Stegocarpous mosses that lack a peristome, e.g., Physcomitrium, are termed gymnostomous. Although they lack a peristome at capsule maturity, such mosses, nonetheless, display characteristic peristomial layers in their developing capsules, and can be aligned with peristomate taxa using their peristomial formulae.
Spores
Most mosses are isosporous, meaning that spore sizes are unimodal, with variation ranging around one arithmetic mean (G. S. Mogensen 1983). Some dioicous mosses that produce dwarf males, however, are anisosporous (D. H. Vitt 1968). In this case, half of the spores in any capsule are significantly smaller than the other half, that is, spore sizes are bimodal within a single capsule (H. P. Ramsay 1979). Culture studies have documented that in many taxa the small spores germinate later than the large spores and give rise to dwarf males (M. Ernst-Schwarzenbach 1944). Bimodality of spore sizes does not, however, always correlate with sexual dimorphism. In some instances, the small spores are consistently abortive, a condition termed pseudo-anisospory (Mogensen 1978). Mogensen hypothesized that a lethal combination of alleles from two genes is responsible for the abortive spores and that this condition leads to balanced polymorphism in the taxon. Pseudoanisopory is more common than true anisospory and occurs in both dioicous and monoicous taxa.
Spores in the majority of mosses are dispersed as single cells, but precociously germinated multicellular spores occur in some xerophytes or epiphytes, such as Drummondia. Spores are typically spheroidal, but may also be ovoid, reniform, or tetrahedral. They are often small, less than 20 µm in diameter, with a finely papillose ornamentation, but much larger, more highly ornamented spores can also occur, primarily in cleistocarpic taxa. Ornamentation of the outer spore wall comes primarily from the perine, which is formed from deposits of globular materials produced within the spore sac (G. S. Mogensen 1983), although in some taxa, e.g., Polytrichum and Astoma, the exine also contributes to the sculpturing (J. W. McClymont and D. A. Larson 1964). In most cases the globular deposits of perine appear to be rather randomly deposited over the spore surface as granulose papillae, e.g., Cinclidium (Mogensen 1981), but in others the deposits seem to be laid down in a regular pattern, perhaps controlled by a predetermined network on the spore surface, e.g., Funaria (H. V. Neidhart 1979). Variations in spore wall ornamentation have been little used in moss systematics, with the exception of a few groups that have been studied using SEM, e.g., Polytrichopsida (Gary L. Smith 1974), Encalyptaceae (D. H. Vitt and C. D. Hamilton 1974), and Pottiaceae (K. Saito and T. Hirohama 1974; J. S. Carrión et al. 1990).
|
|